Neurocognitive consequences of marihuana—a comparison with pre-drug performance
This originally appeared at https://pubmed.ncbi.nlm.nih.gov/15734274/
Abstract
In determining the effects of regular marihuana use on neurocognition, abilities within specific relevant cognitive domains prior to regular drug use have not been available. The present study examined effects of current and past regular use of marihuana in subjects for whom predrug performance had been ascertained in a prospective, longitudinal fashion. A total of 113 young adults, assessed since infancy, were evaluated using neurocognitive tests for which commensurate measures were obtained prior to the initiation of marihuana smoking. Marihuana users, determined by urinalysis and self-report, were categorized as light(b5 joints per week) and heavy (z5 joints per week)current users and former users, the latter having used the drug regularly in the past (z1 joint per week) but not for at least 3 months. A third of the subjects were using marihuana on a regular basis at the time of assessment with half being heavy users. Among former, regular users, approximately half had been smoking 5 or more joints per week. Overall IQ, memory, processing speed, vocabulary, attention, and abstract reasoning were assessed. After accounting for potentially confounding factors and pre-drug performance in the appropriate cognitive domain, current regular heavy users did significantly worse than non-users in overall IQ, processing speed, immediate, and delayed memory. In contrast, the former marihuana smokers did not show any cognitive impairments. It was concluded that residual marihuana effects are evident beyond the acute intoxication period in current heavy users after taking into account pre-drug performance but similar deficits are no longer apparent 3 months after cessation of regular use, even among former heavy using young adults.D 2004 Elsevier Inc. All rights reserved.
Keywords: Marihuana; Cognition; Current use; Former use; Pre-drug performance
1. IntroductionObjective reports examining the presence and nature of a cognitive dysfunction beyond marihuana’s acute intoxication period are equivocal [33,41]. Most studies that have examined heavy marihuana users for possible long-term neuropsychological impairments assessed subjects after an abstinence period of only a day or two [2,3,7,10,34,43]. Among such studies, the areas of vulnerability appear to be memory [43], attention [10,34,43], abstract reasoning [34], and processing speed [10]. As cannabinoid metabolites have been identified in the urine of long-term users for weeks following abstinence [9,11,27], a negative impact observed following recent use may arise from residue of cannabinoids in the central nervous system (CNS) or may reflect withdrawal effects. In either case, any finding may well be temporary and reversible [5,6]. On the other hand, CNS alterations noted in individuals abstinent for a sufficient length of time that impairments cannot be attributed to drug residue [41] raise the specter that the cognitive deficits may be irreversible.Although there is considerable consensus that gross cognitive impairment is not evident in chronic users compared to irregular or non-users following lengthy abstinence periods [13,29,36,40,42], when specific cognitive deficits are considered, the evidence is inconclusive. A recent, quantitative synthesis of research examining various neurocognitive domains found performance in the spheres of learning and memory to be slightly negatively impacted in former chronic users [25]. However, the authors of this
meta-analysis as well as other researchers [19,23,33,42,43] have emphasized that the pre-drug status of individuals is unknown and, without this knowledge, drug effects may be attributable to some preexisting difference(s). Attempts to deal with this issue include matching procedures on [38], or statistically controlling for [34], a variety of non-marihuana variables. There have also been attempts to estimate predrug intellectual functioning by using standardized ability test scores obtained in the fourth grade [2] or contemporary measures of general cognitive functioning that are assumed to be relatively resilient to brain damage [42]. Although these latter approaches are an important improvement, they beg the question of pre-drug functioning in the specific domains of cognitive functioning that appear vulnerable to cannabis use.As well as better ascertainment of pre-drug cognitive functioning [23,33,43], suggestions for improving study designs have emphasized that comparison groups be as similar as possible to drug-using groups [33,34,41,43] and that a prolonged abstinence period is necessary in order to separate drug residual effects from longer lasting neurotoxic CNS effects [23,33]. A longitudinal, prospective design may serve to address these concerns as such a paradigm permits both within- and between-subject comparisons before, during and after quitting regular marihuana use.Recently [19], we reported such an approach investigating IQ scores before, during, and after cessation of regular marihuana use in young adults assessed in a host of neurocognitive measures since birth. Using the difference in age-adjusted IQ scores obtained at ages 9–12 compared to scores of the same individuals at 17–20, current marihuana use had a negative effect on global IQ in subjects who were smoking 5 or more joints a week at the time of the latter assessment. However, what aspects within this global measurement might have contributed to the drug effect was not ascertained. It is important to note that although the heavy users in this predominantly middle-class sample experienced a decrease in IQ scores, their scores were still above population norms and, if the preteen IQ scores were not available, these subjects would have appeared to be unaffected. This negative impact was not noted among users of this amount who had stopped smoking marihuana regularly for at least 3 months.The objective of the present work is to determine whether parallel findings would be observed when specific cognitive functions such as memory, processing speed, abstract reasoning, and attention were evaluated after adjusting for the subjects’ pre-drug performance in each of these domains.
2. Methods
2.1. Participants
The Ottawa Prenatal Prospective Study (OPPS) was initiated in 1978 with the primary objective of examining
the effects on offspring of soft drugs used during pregnancy. In this predominantly middle class, low risk sample, approximately 160 children have been administered neuropsychological tests yearly to age 7 and once during each ofthe 9–12, 13–16, and 17–21 year intervals. The method of recruitment of women early in their pregnancies, the determination of their drug use [17], and a summary of the findings for the children from birth to adolescence have been presented elsewhere [14,15,17,18,20]. In the present study, data from the 9–12 year testing (pre-teen) were used as pre-drug performance measures for the 17–21 year testing(young adult) to evaluate putative changes associated with marihuana use, either current or discontinued.Of the 152 subjects who have been tested between 17 and 21 years of age, 121 were available who had also been tested at 912 years of age. After the test battery was administered to the young adults, additional exclusions were one subject on ritalin, one subject because of amphetamine use, an LSD user, an uncooperative subject, three cases with a positive urinalysis for cannabinoid metabolites coupled with a negative self-report, and one previous regular user who had quit for several months but began regular use a few weeks before testing. The final sample comprised 113 subjects, 50 females and 63 males. No subjects taking psychotropic medications or reporting drug use other than marihuana, cigarettes, or alcohol were included in the analyses.None of the young adult subjects was smoking marihuana regularly at the 9–12 year testing. Of the 113 subjects, 10 had tried marihuana between 9 and 12 years: 1 from the control group and 2, 6, and 1 from the current light, current heavy, and former user groups, respectively.
2.2. Procedures
The tests were administered in laboratories at Carleton University in Ottawa from approximately 9 AM to 3 PM with a supervised lunch break. The early commencement of testing, the reported lack of marihuana use on that day, and the observations of the assessor make it unlikely that subjects were intoxicated during the testing session. Marihuana measures used were the self-reported number of joints smoked per week on a regular (at least 1 joint per week) basis, length of time used, age of smoking onset and the number of joints used over the subject’s lifetime. Urine samples taken on the day of testing of the young adults were used to confirm positive or negative self-reports of marihuana, amphetamines, opiates, and cocaine [32]. Subjects were included only if they reported no drug use other than marihuana, cigarettes, or alcohol and had urinalysis consistent with their self-reports. Marihuana use was categorized into current regular heavy marihuana smokers (z5 joints/wk; n=19), current regular light smokers (b5 joints/wk; n=19), former regular users (no regular use for z3 months and V2 joints in past 2 months; n=16), and a comparison group (never used
marihuana regularly; n=59). The division into light vs. heavy current use at 5 joints per week is consistent with earlier reports [19]. Dividing the creatinine-normalized urinary cannabinoid metabolite levels at the median andexamining concordance with the two group division of current users based on self-report resulted in high concordance [v2(1)=17.8 (pb0.001)] with 84% of both selfreported user groups falling into their corresponding metabolite categories. The Pearson product-moment correlation coefficient was 0.66 (pb0.001) between the reported quantity of marihuana use and the urinary cannabinoid levels. The accepted cutoff value of 50 ng/ml cannabinoid concentration [32] was exceeded by all subjects reporting current heavy use.From the test battery administered to the young adults, only those tests with commensurate measures from the 9 to 12 year testing were analysed. These selected tests covered the cognitive areas of overall IQ, processing speed, memory, vocabulary, facets of attention, and abstractingability (Table 1).
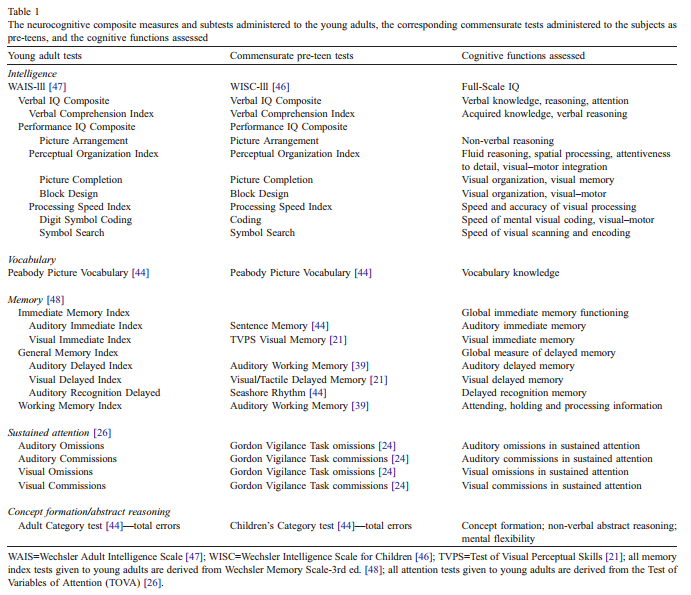
2.3. Statistical analysis
For each cognitive outcome, analysis of covariance (ANCOVA) was conducted to control for pre-drug performance and confounding variables with an a priori contrast comparing each marihuana group (current heavy, current light, and former) with the control group. The analyses were repeated without control for pre-drug performance in order to assess the importance of considering a pre-drug level of performance. Potential confounds considered were SES variables (family income, parental education), maternal use of alcohol, cigarettes, and marihuana during pregnancy, age and sex of subject, young adult’s cigarette and alcohol use, and DSM positive criteria for any of the following DSM-IV Axis I [1] disorders:
generalized anxiety, major depression, dysthymic disorder, attention deficit/hyperactivity disorder, conduct disorder, oppositional defiant disorder, alcohol dependenceand abuse. The computerized National Institute of Mental Health Diagnostic Interview Schedule [8] was used to derive the symptom criteria. Any potential confound that was related to both marihuana use groups with an alpha of 0.1 and the cognitive outcome with an alpha level of 0.05 was controlled for in the analyses [28]. Sex by marihuana use interactions on cognitive outcomes were examined, none was found, anddata are not presented. All analyses were conducted with SPSS version 10.0 (SPSS Institute, Chicago, IL).
3. Results
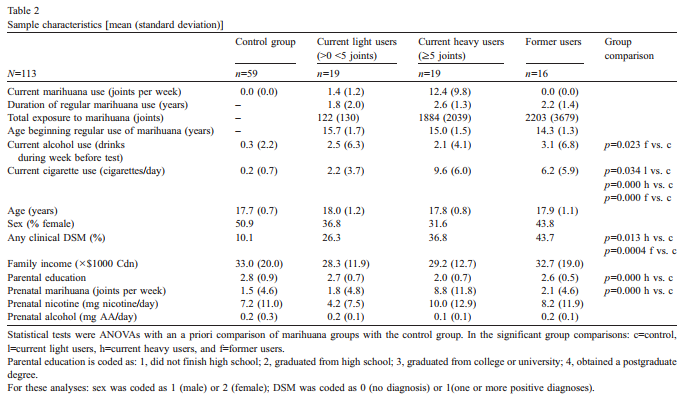
3.1. Sample characteristics
The characteristics of the sample across levels of drug use are described in Table 2.Although on average, the current and former marihuana using groups showed more positive DSM criteria than the comparison group, the frequency of positive diagnoses for each of the disorders considered was low. To use the diagnostic information on non-drug disorders as a covariate, a dichotomous measure was created whereby each subject with one or more positive diagnoses was assigned a d1T or a d0T if no diagnoses. Assumptions of the various analytical procedures were met [45]. Maternal drug use values used as covariates were log-transformed to reduce positive skewness.
3.2. Group differences
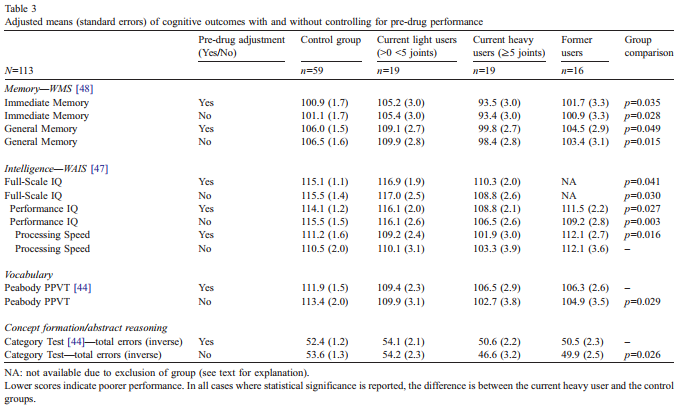
Using four categories of marihuana use in ANCOVA, the current heavy use group but not the former users or current light use group differed from the comparison group inaspects of both memory [48] and IQ [47] (Table 3). After controlling for pre-use performance, the current heavy use group had lower mean scores in Immediate Memory and General (delayed) Memory as well as the Processing Speed Index, a composite score contributing to overall IQ. In addition, the current heavy user group was lower on FullScale IQ compared to controls after the former user group was omitted due to a factor by covariate interaction that violated the assumption of homogeneity of regression coefficients. Compared with the control group, the former use group had poorer predicted adult IQ scores at the lower preteen IQ level but elevated predicted adult IQ scores at the higher preteen IQ level. Dividing the preteen IQ scores at 110 yielded approximately equal groups and resulted in a lack of interaction for the lower preteen scores. However, the higher preteen scores continued to display a significant interaction and, with too few subjects scoring above 110, further study of the interaction was not possible. Using the lower preteen scores that exhibited no interaction, an ANCOVA comparing the former use group with the control group revealed no significant difference in young adult IQ [adjusted means (S.E.): 104.3 (3.3) and 107.9 (1.6), respectively].A measure of incidental learning [49], the digit symbol task of the WAIS [47], was examined to determine if failure to learn or remember digit/symbol combinations mediated the relationship between Processing Speed and current heavy marihuana use. After using the incidental learning measure as an additional covariate, the previous statistically significant relationship was retained. After controlling for pre-drug performance, no relationships were found between current marihuana use and working memory [48], Verbal IQ [47], the Peabody Picture Vocabulary Test (PPVT) [44], the Category Test [44], or the TOVA attention tasks [26]. The lack of a predictive association between marihuana use and the TOVA visual tasks contrasts with the significant association of marihuana use found with the visual-based Processing Speed Index. The possible commonality of these visual measures was examined by Pearson product-moment correlations using the comparison group. The correlation coefficients between each of the TOVA visual tasks and Processing Speed were low and non-significant, indicating that these measures assess relatively independent neurocognitive elements. Attention in both auditory and visual modalities utilizing the TOVA was examined with the target stimulus occurring at high and low probability levels but examination of these subscales revealed no differential association with current marihuana use.
3.3. Potential influences on marihuana–outcome relationships Controlling for pre-drug performance resulted in changes in statistical significance for several cognitive outcomes(Table 3). Processing Speed Index [47] was associated with current heavy marihuana use after, but not before, pre-drug performance control. Conversely, both the PPVT [44] and Category Test [44] were associated with current heavy marihuana use before, but not after, pre-drug performance control. The impact of both recency of use and positive clinical diagnosis on the relationships found between marihuana use and cognitive variables was examined. Self-reports of marihuana usage in each of the 7 days previous to testing, when individually correlated with relevant outcomes, did not reveal stronger negative associations with more recent exposure. The role of clinical diagnosis was examined in all analyses by removing it as a covariate and noting whether a change in level of statistical significance resulted. In no cases did the DSM [1] score mediate the relationship between the outcomes and marihuana usage. Two subjects had tried marihuana on an irregular basis before their 9–12 test date. When these two subjects were excluded from the analyses, no changes were noted in any of the previously statistically significant relationships.
3.4. Former users In contrast to the current heavy marihuana users, the former users, as a whole, did not differ from the comparison group. This group included nine subjects who had smoked fewer than 5 joints per week and seven subjects who had smoked at least 5 joints per week. Both the former heavy users and the current heavy users smoked marihuana regularly for the same length of time (2.8 years), but the former user group started regular use slightly younger (14.3 years) than the current heavy users (15 years), and consumed a total of approximately 5000 joints compared with 2000 for the current users. The analyses were repeated to determine if the former heavy using group was different from the comparison group on any of the cognitive outcomes and no significant differences were noted.
4. Discussion
The results of this study are consistent, in their broad strokes, with much of the extant literature but important differences and extensions exist due to both the longitudinal design of the work permitting the use of pre-drug controls and the nature of many of the outcomes assessed. The present work is the first in which the actual domains measured in young adult current and former users were also evaluated prior to the subjects’ onset of regular cannabis use.The importance of evaluating pre-drug functioning within specific cognitive domains is clearly evident in the assessment of the impact of current marihuana use in the Category Test [44] and the PPVT [44]. If the level of performance on these tests prior to marihuana use had not been taken into account, an inappropriate attribution of deficits in these outcomes to current marihuana smoking would have occurred. The value of pre-drug control is evident not only with qualifying these previously significant associations, but also unmasking previously non-significant statistical relationships. For example, without pre-drug control, no impact of current heavy marihuana use on the Processing Speed Index [47] would have been noted.Consistent with our earlier report [19] the IQ of current heavy users was significantly lower than the control group. That this observation was noted after pre-drug general intelligence was used as a statistical control is an important addition to the extant literature. Visual processing speed, one of the major composite index scores contributing to the IQ measurement, appeared to be the most vulnerable domain. This putative impact of marihuana use on visual processing is consistent with another study in which cannabis-using subjects performed worse in a task requiring the rapid scanning and identification of visual stimuli [10].In the current report, processing speed was assessed solely in the visual domain and this may be important. In the one other report in which information processing was assessed in heavy marihuana users, only the auditory modality was examined and mixed results were noted [43] with marihuana smokers being impaired on verbal processing but not in a serial addition task. An interpretation might be that the language information processing task utilizes visual imagery to a greater degree than does resolution of the mathematical problems and that deficits in visualprocessing, as described above, may negatively impact on visual imagery. This interpretation is consistent with the observation on a word recall memory task in which a retrieval deficit among marihuana users was restricted towords that were easy to image visually [2]. It was speculated by the authors that such an impairment might reflect a slowed processing of the visual representation of the word. One of the strongest negative associations with current heavy regular use among the neurocognitive domains examined was in the area of memory, both immediate and delayed. These observations are consistent with and extendthe findings of the meta-analytic report by Grant et al. [25]. In that overview, it was concluded that a small but statistically significant effect was associated with marihuana use although these authors tempered their observations by emphasizing that the pre-drug cognitive abilities of the subjects were largely unknown. The present findings address this valid concern.That young adult, current heavy regular cannabis users performed significantly worse in the areas of visual processing and aspects of memory is congruent with the fact that specific cannabinoid receptors, through which the drug exerts its effect, are highly expressed in the hippocampus [22]. This structure plays an important role in memory and is of considerable importance in visual–spatial learning [31]. Thus, alterations of hippocampal function induced by chronic marihuana [30] may underlie, at least in part, the subtle neurocognitive changes noted in the heavy users.Only a limited number of reports have utilized paradigms designed to examine attentional functions in tasks that minimize other facets of cognitive functioning among marihuana users. The results of these studies are inconsistent. For example, marihuana users in their early 20s differed from a non-using control group in a divided attention task failing to improve reaction time to a visual stimulus with the presence of a preceding acoustical signal. However, reaction time without the preceding signal and, in a further paradigm, the ability to shift the focus of attention were not impaired [10]. In another report that, as in the present study, employed a continuous performance paradigm, no association among 30–55 year olds with drug use was reported in error rates in either a visual or auditory continuous performance task when the subjects were assessed 1 and 7 days after abstaining from drug use [35]. The nature of the errors considered was not specified. These findings are consistent with those of the present work with young adults in which errors of omission and commission were not significantly related to marihuana use in either a visual or auditory modality. Controlling for pre-drug performance did not alter observations in either modality.It is apparent that the relationship between current marihuana use and attention is far from clear, reflecting the multifaceted nature of this cognitive domain [10,16,35]. The failure to find an impact of regular cannabis use on the visual continuous performance task in the present study appears at odds with the impact of the drug on the various other cognitive domains in which visual processing is a vulnerable aspect. However, the deficits in visual information processing are largely independent of competency in visual attention as demonstrated both by the low correlations between the visual attention task and the Processing Speed Index in the present work as well as with Wechsler’s report of only low to moderate correlations between several measures of attention and the Visual Processing composite score [49].Cannabinoid receptor densities vary in different parts of the brain during developmental stages and thus the age of chronic exposure is, potentially, an important factor in evaluating the consequences of marihuana. Researchers [42] have speculated that adolescent users are at increased risk for negative outcomes. Impaired reaction times in a visual scanning task were noted among young adults if the onset of regular use was before but not after the age of 16 [10]. As regular use of marihuana was initiated during mid- to late adolescence among both the current and former users in the present work, the possible contributing role of this age factor should be recognized. The observations in the present study were made when the current heavy users were not intoxicated but typically had smoked marihuana within the preceding day or two and had concentrations of cannabinoid metabolites above standard cutoff values [32] in their urine. It is unlikely that the impairments noted were due to withdrawal [6] as the subjects were not asked to vary their normal pattern of marihuana consumption, as no change in the effects as a function of recency of use was observed, and as currently using subjects did not self-report irritation, agitation or subjective signs of withdrawal on the clinical questionnaire [8].In contrast to the effects of cannabis detected within a few days of marihuana use, neurocognitive performance among former light and heavy users showed no significant, negative effect across all cognitive domains examined including those that were performed poorly by the current users. This observation was not altered by the inclusion of pre-drug performance. These findings among the former users are consistent with those of Pope [35,36] in which 30– 55 year olds who had smoked marihuana at least 5000 times and who were smoking daily at the time of entry into the study were assessed after a 0-, 1-, 7-, and 28-day abstinence period. Memory deficits noted in the first three testing periods had almost completely disappeared by the 28th day. However, recently, Pope et al. [37] upon subdividing this sample into those who started smoking before the age of 17 and those who began at 17 or older reported that the early onset group exhibited poorer cognitive performance than later-onset or control subjects after the 28-day abstinent period, particularly in tasks involving verbal abilities. The researchers cautioned that unmeasured pre-drug differences may underlie the age-related findings. In another study [4], also using a 28-day abstinent period, it was reported that seven individuals averaging 94 joints per week scored worse than seven users averaging 11 joints per week on a number of memory tests as well as aspects of executive function, psychomotor speed and manual dexterity. However, as there was no control group or baseline measures with testing occurring only after the 1-month abstinence, interpretation of the deficits among the very heavy users is problematic. Whether these differences persisted beyond this period was not examined. In our study, lack of regular use by the former users was validated by urinalysis for the few days before testing and by self-report for the previous 3 months. The absence of a negative impact among the former heavy users in the present report is all the more striking as they initiated their regular habits at a younger age than the current users (14 vs. 15 years). In fact, at the time of assessment, the total number of joints smoked in the lifetime of the young adults was estimated to be approximately 4800 among the former heavy users in contrast to the approximate 1900 among the current heavy users. The cognitive impairments among the current heavy users and the recovery of function among the heavy, former users do not appear to have clinical underpinnings. Although the marihuana groups manifested a greater incidence of positive diagnosis on the clinical outcomes examined, use of this variable as a control did not affect the presence or absence of statistical significance. Within the parameters examined in the present work, the absence of significant cognitive differences between the former users and the control group suggests that residual marihuana-associated deficits observed among current users are transient phenomena and that regular, heavy use does not produce irreversible, neurocognitive effects. However, caution must be exercised in interpreting and generalizing the results obtained from the former heavy users in the present work. The average length of time of regular use by these former smokers was, like the current users, slightly over 2 1/2 years. This is a relatively short time and consideration must be given to the possibility that if marihuana had been used for a longer duration prior to cessation, reversal of cognitive effects may not have occurred. However, the evidence on this issue is inconclusive with some workers reporting an association between life-time duration of former use and cognitive functioning[12,40,43] while others have found no such relationship [4,29,36].In summary, this is the first study to use a prospective, longitudinal approach in which domain specific pre-drug cognitive performance levels were considered in evaluating the effects among users and former users. Significant consequences of heavy, current regular marihuana use were noted among the young adults in intelligence, memory, and speed of visual processing but this negative impact was no longer evident among individuals who had ceased regular smoking for 3 months. By using tests assessing specific functioning before and after the initiation of regular marihuana smoking, greater confidence is engendered in the interpretation of the drug’s effects in current and former users.
References
[1] American Psychiatric Association, Diagnostic and Statistical Manual of Mental Disorders, 4th ed., American Psychiatric Association, Washington, 1994, DSM-IV.[2] R.I. Block, M.M. Ghoneim, Effects of chronic marijuana use onhuman cognition, Psychopharmacology 110 (1993) 219–228.
[3] R.I. Block, S. Farnham, S. Braverman, R. Noyes Jr., M.M. Ghoneim,Long-term marijuana use and subsequent effects on learning and cognitive functions related to school achievement: preliminary study, NIDA Res. Monogr. Ser. 101 (1990) 96– 111.
[4] K.I. Bolla, K. Brown, D. Eldreth, K. Tate, J.L. Cadet, Dose-relatedneurocognitive effects of marijuana use, Neurology 59 (2002)1337– 1343.[5] A.J. Budney, J.R. Hughes, B.A. Moore, P.L. Novy, Marijuanaabstinence effects in marijuana smokers maintained in their home environment, Arch. Gen. Psychiatry 58 (2001) 917–924.
[6] A.J. Budney, B.A. Moore, R.G. Vandrey, J.R. Hughes, The timecourse and significance of cannabis withdrawal, J. Abnorm. Psychology 112 (2003) 393– 402.
[7] L.D. Chait, Subjective and behavioral effects of marijuana themorning after smoking, Psychopharmacology 100 (1990) 328– 333.
[8] Computerized NIMH-Diagnostic Interview Schedule for Childrenversion IV (C-DISC 4), Columbia University, New York, 1997.
[9] J.S. Cridland, D. Rottanburg, A.H. Robins, Apparent half-life ofexcretion of cannabinoids in man, Human Toxicol. 2 (1983) 641–644.[10] H. Ehrenreich, T. Rinn, H.J. Kunert, M.R. Moeller, W. Poser, L.Schilling, G. Gigerenzer, M.R. Hoehe, Specific attentional dysfunction in adults following early start of cannabis use, Psychopharmacology 142 (1999) 295– 301.
[11] G.M. Ellis Jr., M.A. Mann, B.A. Judson, N.T. Schramm, A. Taschian,Excretion patterns of cannabinoid metabolites after last use in a group of chronic users, Clin. Pharmacol. Ther. 38 (1985) 572– 578.
[12] J.M. Fletcher, B. Page, D.J. Francis, K. Copeland, M.J. Naus, C.M.Davis, R. Morris, D. Krauskopf, P. Satz, Cognitive correlates of longterm cannabis use in Costa Rican men, Arch. Gen. Psychiatry 53(1996) 1051– 1057.[13] P.A. Fried, Commentary 2, Evid.-Based Public Health 6 (2002)109– 110.
[14] P.A. Fried, Conceptual issues in behavioral teratology and theirapplication in determining long-term sequelae of prenatal marihuana exposure, J. Child Psychol. Psychiatry 43 (2002) 81–102.
[15] P.A. Fried, Adolescents prenatally exposed to marijuana: examinationof facets of complex behaviors and comparison with the influence of in utero cigarettes, J. Clin. Pharmacol. 42 (2002) 97S– 102S.
[16] P.A. Fried, B. Watkinson, Differential effects on facets of attention inadolescents prenatally exposed to cigarettes and marihuana, Neurotoxicol. Teratol. 23 (2001) 421– 430.
[17] P.A. Fried, B. Watkinson, A. Grant, R.K. Knights, Changing patternsof soft drug use prior to and during pregnancy: a prospective study, Drug Alcohol Depend. 6 (1980) 323–343.
[18] P.A. Fried, B. Watkinson, R. Gray, Differential effects on cognitivefunctioning in 9- to 12-year olds prenatally exposed to cigarettes and marihuana, Neurotoxicol. Teratol. 20 (1998) 293– 306.
[19] P. Fried, B. Watkinson, D. James, R. Gray, Current and formermarijuana use: preliminary findings of a longitudinal study of effects on IQ in young adults, Can. Med. Assoc. J. 166 (2002) 887–891.
[20] P.A. Fried, B. Watkinson, R. Gray, Differential effects on cognitivefunctioning in 13- to 16-year olds prenatally exposed to cigarettes and marihuana, Neurotoxicol. Teratol. 25 (2003) 427– 436.
[21] M.F. Gardner, TVPS Test of Visual–Perceptual Skills, Special ChildPublications, Seattle, WA, 1982.
[22] M. Glass, M. Dragunow, R.L.M. Faull, Cannabinoid receptors in thehuman brain: a detailed anatomical and quantitative autoradiographicstudy in the fetal, neonatal and adult human brain, Neuroscience 77(1997) 299– 318.
[23] R. Gonzalez, C. Carey, I. Grant, Nonacute (residual) neuropsychological effects of cannabis use: a qualitative analysis and systematic review, J. Clin. Pharmacol. 42 (2002) 48S– 57S.
[24] M. Gordon, D.F. McClure, Gordon Diagnostic System: InterpretativeSupplement, Clinical Diagnosis, Golden, CO, 1984.
[25] I. Grant, R. Gonzalez, C.L. Carey, L. Natarajan, T. Wolfson, Nonacute (residual) neurocognitive effects of cannabis use: a metaanalytic study, J. Int. Neuropsychol. Soc. 9 (2003) 679– 689.
[26] L. Greenberg, T.O.V.A. Tests of Variables of Attention, UniversalAttention Disorders, Los Alamitos, CA, 1996.
[27] M.A. Huestis, J.M. Mitchell, E.J. Cone, Detection times of marijuanametabolites in urine by immunoassay and GC-MS, J. Anal. Toxicol.19 (1995) 443– 449.
[28] J.L. Jacobson, S.W. Jacobson, Methodological issues in humanbehavioral teratology, in: C. Rovee-Collier, L.P. Lipsitt (Eds.),Advances in Infancy Research, vol. 6, Ablex, Norwood, NJ, 1990.
[29] C.G. Lyketsos, E. Garrett, K.Y. Liang, J.C. Anthony, Cannabis useand cognitive decline in persons under 65 years of age, Am. J.Epidemiol. 149 (1999) 794– 800.
[30] G. Mereu, M. Fa, L. Ferraro, R. Cagiano, T. Antonelli, M. Tattoli, V.Ghiglieri, S. Tanganelli, G.L. Gessa, V. Cuomo, Prenatal exposure to a cannabinoid agonist produces memory deficits linked to a dysfunction in hippocampal long-term potentiation and glutamate release, Proc. Natl. Acad. Sci. 100 (2003) 4915– 4920.
[31] J. O’Keefe, L. Nadel, The Hippocampus as a Cognitive Map, OxfordUniversity Press, Oxford, 1978.[32] M.R. Pearce, L.D. Tarnai, A. Poklis, Performance evaluation of fouron-site drug-testing devices for detection of drugs of abuse in urine, J. Anal. Toxicol. 24 (2000) 589–594.
[33] H.G. Pope Jr., A.J. Gruber, D. Yurgelun-Todd, The residual neuropsychological effects of cannabis: the current status of research, Drug Alcohol Depend. 38 (1995) 25–34.
[34] H.G. Pope Jr., D. Yurgelun-Todd, The residual cognitive effects ofheavy marijuana use in college students, JAMA 275 (1996) 521– 527.
[35] H.G. Pope Jr., A.J. Gruber, J.I. Hudson, M.A. Huestis, D. YurgelunTodd, Neuropsychological performance in long-term cannabis users, Arch. Gen. Psychiatry 58 (2001) 909– 915.
[36] H.G. Pope Jr., A.J. Gruber, J.I. Hudson, M.A. Huestis, D. YurgelunTodd, Cognitive measures in long-term cannabis users, J. Clin.Pharmacol. 42 (2002) 41S– 47S.
[37] H.G. Pope Jr., A.J. Gruber, J.I. Hudson, C. Cohane, M.A. Huestis, D.Yurgelun-Todd, Early-onset cannabis use and cognitive deficits; what is the nature of the association? Drug Alcohol Depend. 69 (2003) 303– 310.
[38] R.H. Schwartz, P.J. Gruenewald, M. Klitzner, P. Fedio, Short-termmemory impairment in cannabis-dependent adolescents, Am. J. Dis. Child. 143 (1989) 1214–1219.
[39] L.S. Siegel, E.B. Ryan, The development of working memory innormally achieving and subtypes of learning disabled children, Child Dev. 60 (1989) 973– 980.
[40] N. Solowij, Cannabis and Cognitive Functioning, Cambridge University Press, Cambridge, 1998.
[41] N. Solowij, Long-term effects of cannabis on the central nervoussystem, in: H. Kalant, W. Corrigall, W. Hall, R. Smart (Eds.), The Health Effects of Cannabis, Addiction Research Foundation, Toronto, ON, 1999, pp. 195– 265.
[42] N. Solowij, B.F.S. Grenyer, Long-term effects of cannabis onpsyche and cognition, in: F. Grotenhermen, E. Russo (Eds.), Cannabis and Cannabinoid, Pharmacology, Toxicology and Therapeutic Potential, Haworth Integrative Healing Press, New York, 2002, pp. 299– 311.
[43] N. Solowij, R.S. Stephens, R.A. Roffman, T. Babor, R. Kadden, M.Miller, K. Christiansen, B. McRee, J. Vendetti, Cognitive functioning
of long-term heavy cannabis users seeking treatment, JAMA 287(2002) 1123– 1131.
[44] O. Spreen, E.A. Strauss, Compendium of Neuropsychological Tests,Oxford University Press, Oxford, 1991.
[45] B. Tabachnick, L.S. Fidell, Using Multivariate Statistics, Harper &Row, New York, 1983.
[46] D. Wechsler, Wechsler Intelligence Scale for Children, 3rd ed., ThePsychological, New York, 1991.
[47] D. Wechsler, Wechsler Adult Intelligence Scale, 3rd ed., ThePsychological, New York, 1997.
[48] D. Wechsler, Wechsler Memory Scale, 3rd ed., The Psychological,New York, 1997.
[49] D. Wechsler, WAIS-lll-WMS-lll Technical Manual, The Psychological, New York, 1997.
